Pathological bone fracture
Pathological bone fracture. The simplified designation of “pathological fracture” is not appropriate, as every fracture is a pathological process. It is correct to use the term: pathological bone fracture, which is most often related to neoplasms, whether primitive or metastatic.
The pathological processes that can lead to fractures are classified as bone dysplasias, circulatory disorders, degenerative, inflammatory and infectious or neoplastic changes.
For the correct diagnosis, it is necessary to consider the patient’s clinical aspects, fracture mechanisms, imaging, laboratory and anatomopathological aspects.
Bone fractures, which hide undiagnosed pathological processes, can result in inadequate orthopedic management.
We will organize them didactically within the five chapters of General Pathology, namely: dysgenesis or dysplasias, degenerative processes, circulatory disorders, inflammations and neoplastic diseases.
BONE DYSPLASIAS:
– dis (from the Greek = alteration), plasien (= form). Any change in bone morphology, whether congenital or hereditary, can cause deformities and/or fractures. Due to the frequency and polymorphism of the anatomical changes they present, we highlight the following:
1 – Osteopsatirosis or Osteogenesis imperfecta:
In any of its manifestations, in Rubin’s classification, it is a hereditary disease that predominates in the diaphysis of long bones and determines changes in bone morphology due to deficient sub-periosteal bone apposition. Longitudinal bone growth occurs at the level of the epiphyseal line, where cartilage transforms into bone tissue. Transverse growth, however, depends on endosteal resorption and subperiosteal bone apposition. Failure of this modeling mechanism in bones leads to impaired growth in the transverse direction. Due to this pathogenesis, the bones become very thin and fragile, subject to frequent fractures.
2 – Osteopetrosis or Albers Schomberg Disease:
It is a disease characterized by changes in the epiphyseal line of bones of endochondral origin. The lesions are condensing due to the failure in the activity of osteoclasts which, under normal conditions, act in the physiological resorption of bones. With apposition prevailing over resorption, the bones initially condense in the metaphysis and, progressively, throughout the entire bone, whose consistency becomes stony. In addition to anemia, which results from the reduction and even absence of marrow spaces, the seat of hematopoiesis, denser bones lose their elasticity and can fracture.
3 – Fibrous dysplasia, mono or polyostotic:
It is a condition in which there is partial replacement of the bone by fibrous proliferation between osteoid beams with little mineralization and has lower radiographic density. With growth and skeletal maturation, progressive ossification generally occurs, which may even resemble normal bone structure. The injured area has a lower density than that of normal bone and, therefore, the main anatomical manifestation is deformity, which sometimes leads to fracture.
METABOLIC CHANGES :
For bones to maintain a normal structure, the apposition and reabsorption mechanisms must be in balance. Apposition depends on the activity of osteoblasts that elaborate the
collagen fibers, protein matrix of bones. Along the collagen fibers there will be deposition of minerals, tricalcium phosphate, in the form of hydroxyapatite crystals. Protein collagen fibers account for 95% of the structure of the bone matrix. The remaining 5% are mucopolysaccharides, hyaluronic and chondroitinsulfuric acid, which predominate in the “cement lines” or “reverse lines”, which delimit the different bands of matrix apposition, maintaining normal bone growth. Simultaneously, bone resorption is carried out by osteoclasts under stimulation of parathyroid hormone. These mechanisms of apposition and reabsorption, which represent the so-called bone remodeling ( turnover ), are intense in the first decade of life, less in the second and progressively less with advancing age, but always present throughout our lives.
Normal bone metabolism, therefore, consists of: a- apposition of the protein matrix whose collagen fibers are produced by osteoblasts, which require muscular activity to fulfill their functions; b- dietary protein intake, vitamins A and C, minerals mainly calcium and phosphorus; c- gonad, thyroid, pituitary and adrenal hormones are also necessary for matrix formation and mineralization. For reabsorption, osteoclasts produce enzymes that enable the dissolution of the matrix and the solubilization of minerals, which act under the stimulus of parathyroid hormone.
Changes in any of the elements that contribute to altering turnover will lead to metabolic bone disease, especially the following:
1 – Osteoporosis:
It is an important and frequent cause of fractures, caused by reduced bone consistency due to the quantitative reduction of the matrix, reducing the mineral deposit bed that leads to greater bone fragility and fractures, especially of the vertebrae and femur. Osteoporosis does not depend on a lack of calcium or phosphates, as it means matrix deficiency, which reduces the area of mineral apposition. The causes arise from less muscular activity in people with a sedentary lifestyle, particularly the elderly or in patients who have been bedridden for a long time, hence the increasing importance of exercise to treat it, in addition to a diet with adequate protein intake. States of protein deficiency due to dietary deficiency or excess elimination are subject to osteoporosis, as occurs in states of malnutrition and diseases with dysproteinemia, such as multiple myeloma and bone carcinomatosis. Osteoporosis also occurs in changes in endocrine glands, such as postmenopausal hypoestrogenism, hyperthyroidism, pituitary adenomas, gigantism and acromegaly, changes in the adrenal cortex, which lead to Cushing’s syndrome, and others.
2 – Osteomalacia and rickets:
These are diseases resulting from mineral deficiency, that is, they do not depend on changes in the protein matrix. In rickets, mineral deficiency predominates in the epiphyseal lines or growth plates, where the demand is greatest and necessary for the mineralization of the newly formed osteoid beams. Due to the lower resistance of these regions, there will be a “cup-shaped” enlargement in the metaphyses of the long bones and a “rickety rosary” in the ribs. Osteomalacia is also known as “adult rickets”. Although uncommon, it translates into generalized mineral deficiency, as there is no growth plate in the bones. Occurs due to reduced intestinal absorption
in patients who have undergone major intestinal resections or due to dietary deficiency. Fractures result from greater bone fragility caused by mineral deficiency.
3 – Hyperparathyroidism:
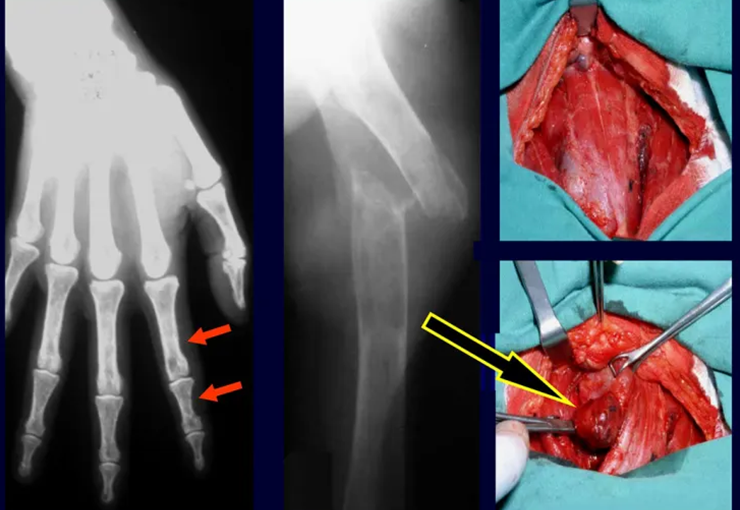
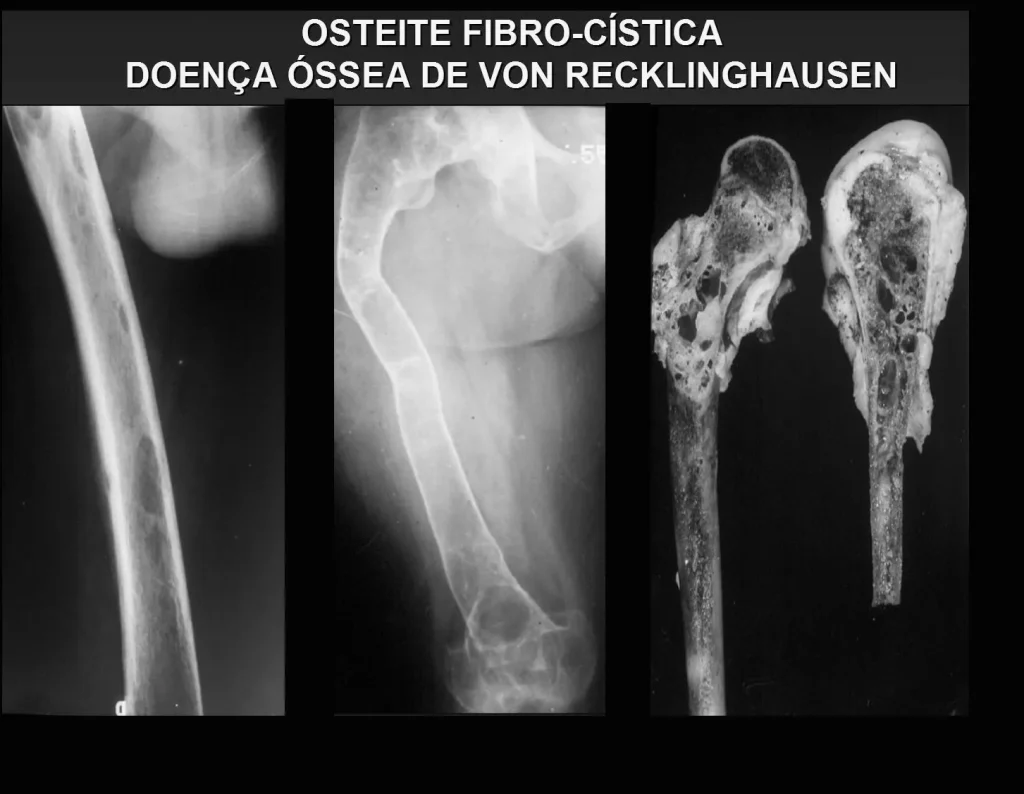
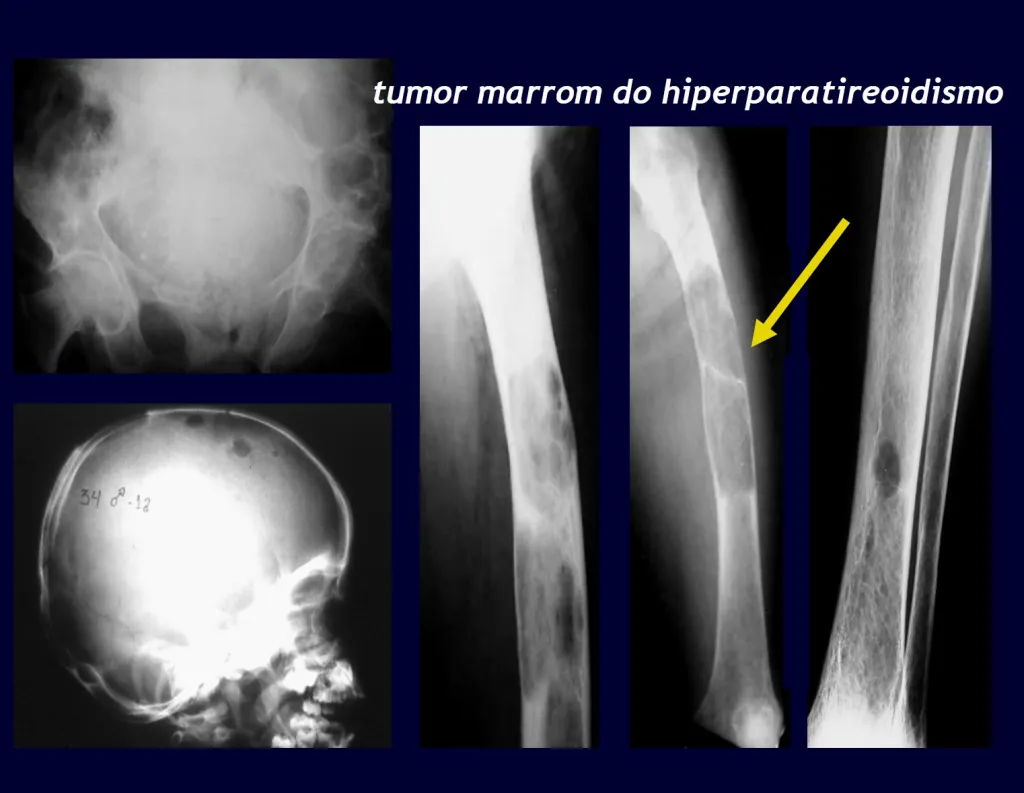
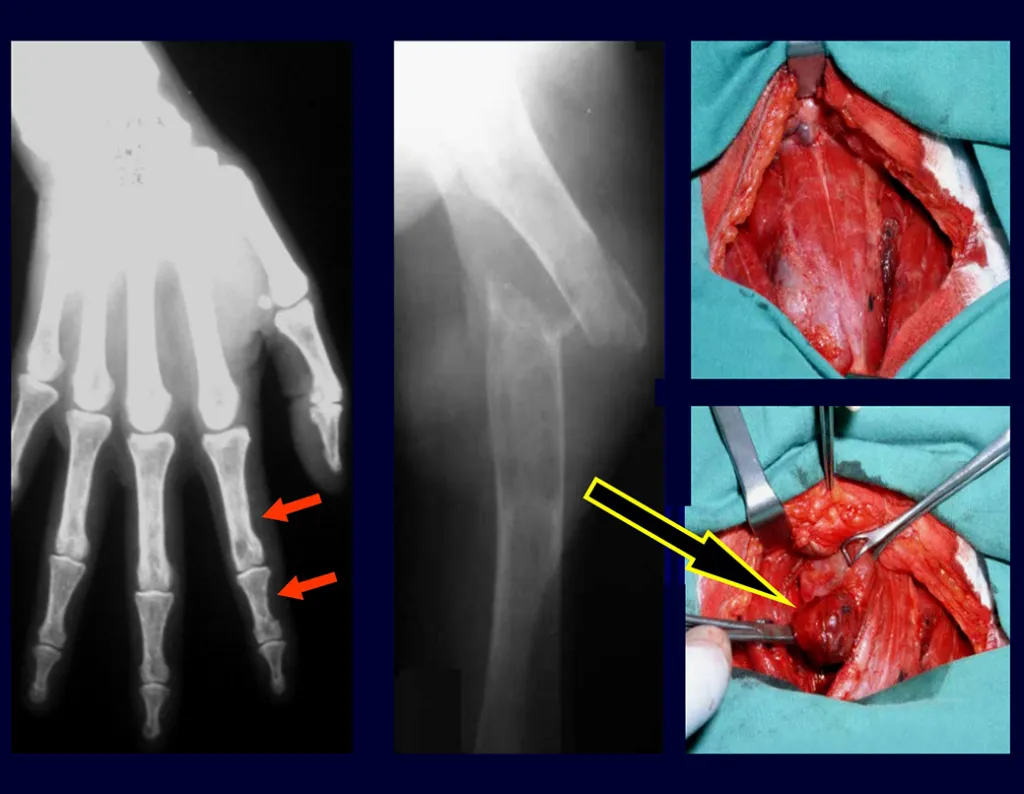
An important cause of pathological bone fracture, often the initial sign of this disease, especially in its primary form, the cause of which is the adenoma of one of the parathyroid glands. Parathormone normally acts on osteoclasts, cells that carry out bone reabsorption. It also acts on the kidneys, inhibiting the tubular reabsorption of phosphates and, in this way, exerts control over phosphaturia and, consequently, over phosphaemia. When there is an excess of parathyroid hormone, there will be hyperphosphateria, altering the Ca/P balance which, under normal conditions, maintains a 2:1 ratio, from the blood (9.5 calcium / 4.5 mg phosphorus) to the hydroxyapatite formula. There will, therefore, be hypercalcemia to maintain blood Ca/P balance. Calcium is removed from the bones, which are the largest depository of this mineral in our body, normally retaining around 95% of this mineral. As a consequence of this process, the bones will become more fragile with spontaneous fractures or due to mild trauma. Another important sign of the disease is recurrent calculosis, especially kidney stones. Primary hyperparathyroidism is a long-term chronic disease that, if not treated with extirpation of the parathyroid adenoma, will fatally lead to progressive and generalized demineralization of the bones with multiple fractures and intraosseous cystic formations, due to the intensity of reabsorption by osteoclasts. Furthermore, the presence of so-called “brown tumors”, isolated or multiple, is common, the pathogenesis of which is due to intraosseous hemorrhages with the presence of ferric hemosiderin pigment, in addition to clusters of osteoclasts. All of these bone changes result in demineralization of the beams and partial replacement by fibrosis, progressing to the so-called generalized fibrocystic osteitis or Von Recklinghausen’s disease of the bones, which should not be confused with neurofibromatosis, which also bears the name of this author.
DEGENERATIVE DISEASES:
Pathological bone fracture
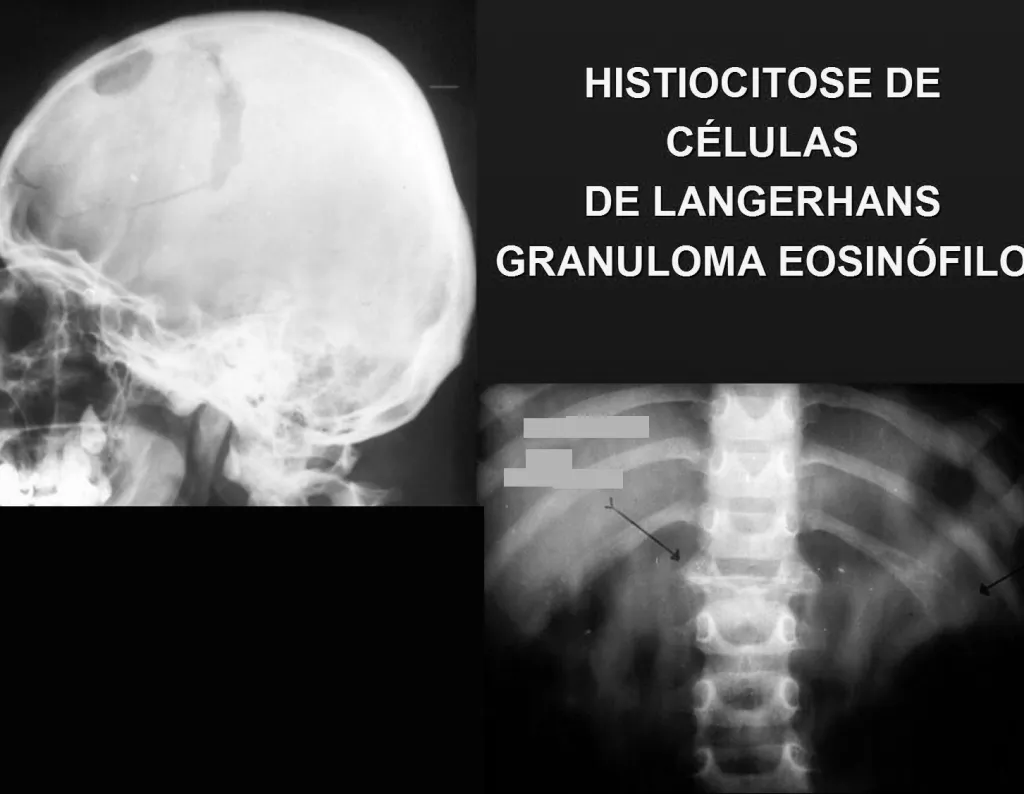
Within this chapter we can include Langerhans cell histiocytoses , called Histiocytoses X, by Lichtenstein, and lipidoses.
1 – Langerhans cell histiocytosis:
-including eosinophilic granuloma and Hand Schiller-Christian disease.
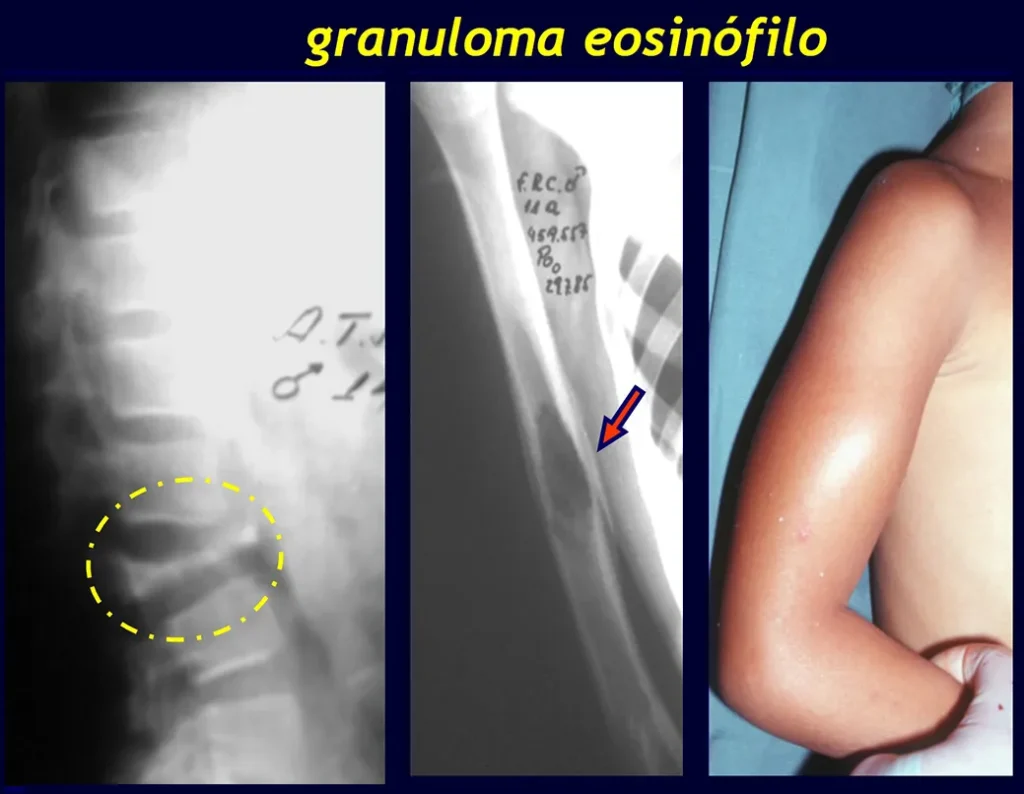
Eosinophilic granuloma:
The most frequent form is Eosinophilic Granuloma , which is more common in children, is generally monostotic and is characterized by an osteolytic lesion in the cranial vault in the form of a circular “bite-shaped” lesion, in the vertebral body and in the diaphysis of long bones. When located in the vertebra, it compromises the body of this bone, with osteolysis and “collapse”, flattening the vertebral body, which constitutes a fracture with radiographic appearance of the so-called flat vertebra of Calvè. In long bones, it affects the diaphysis and, depending on the size of the lesion, fractures may occur.
Hand – Schuller – Christian disease:
In Hand – Schuller – Christian disease , which may be the evolution of eosinophilic granuloma, the lesions are multiple with severe bone involvement, due to the clusters of macrophages that are frequently xanthomylated, due to the accumulation of cholesterol esters. These accumulations can also compromise the pituitary bed and the retro-ocular region, which can lead to the symptomatic triad, that is, multiple osteolytic lesions, diabetes insipidus and unilateral or bilateral exophthalmos. Long bones are often the site of fractures.
2 – Gaucher disease:
Among lipidoses, it is the form that most compromises bones. In this entity, histiocytic cells, due to an enzymatic defect, are full of lipids that replace the structure of the bones, especially in the femurs, constituting an important cause of necrosis of the head of this bone, accompanied by deformities, which can lead to fractures.
CIRCULATORY DISORDERS :
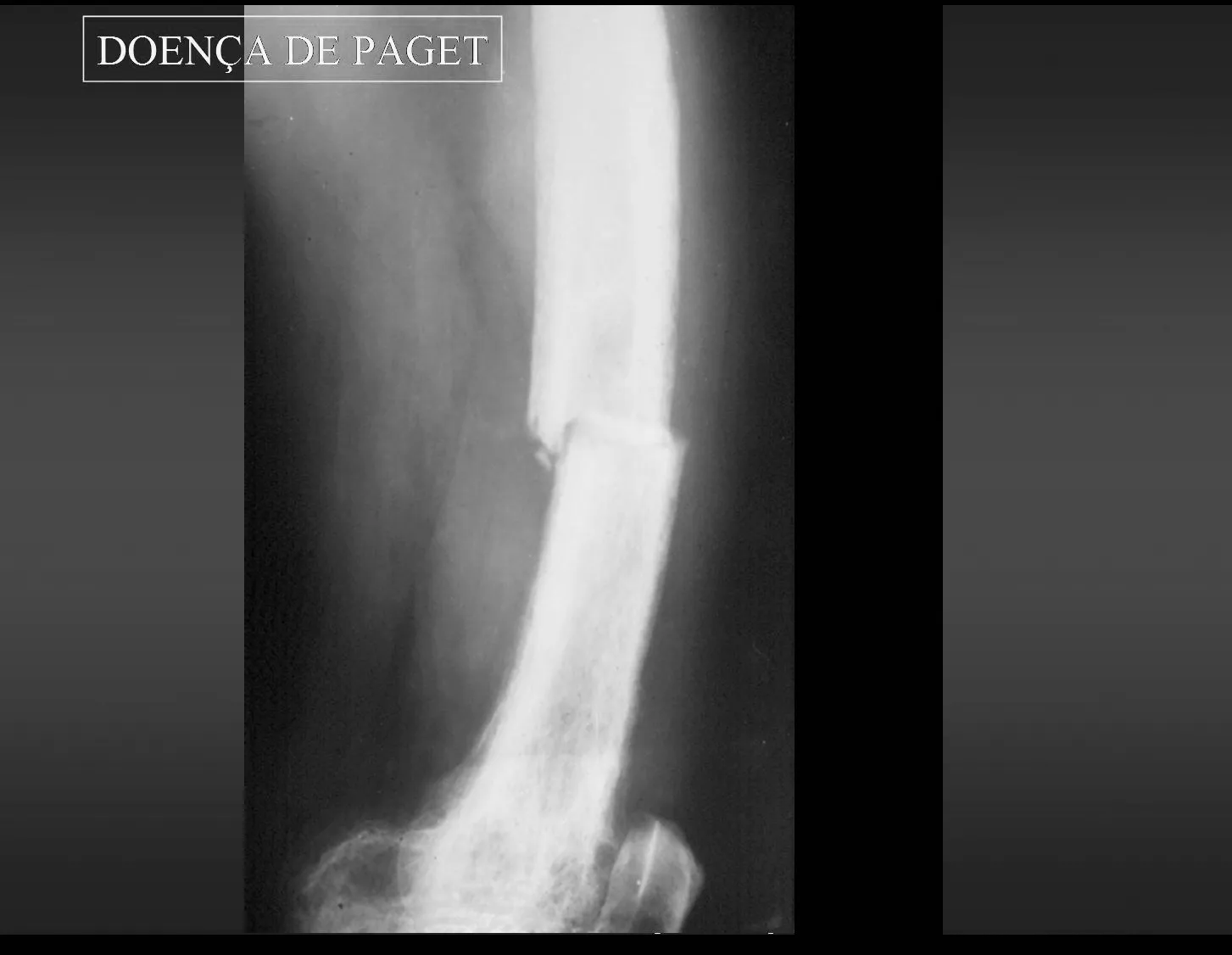
In bone pathology, the most significant example of intraosseous blood circulation disorder occurs in Paget’s disease, also known as osteitis deformans, described in 1892(), by Sir Thomas Paget, and until today considered to be of unknown cause.
1 – Paget’s disease:
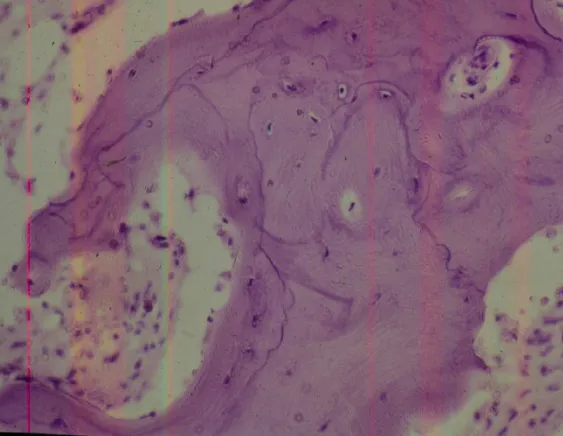
Paget’s disease occurs in advanced age groups, generally over 50 years of age, mono or polyostotic. In the initial stages of this disease, there is a considerable increase in intraosseous arterial circulation which, as it is active, arterial, there is marked bone reabsorption with radiological and anatomopathological lesions that present characteristics of the so-called “circumscribed osteoporosis”, more frequent in the skull, pelvis, femur and tibia. At this stage, fractures may occur due to the greater fragility of the bones. The most frequent symptoms are pain and discomfort in the affected area. Some authors report cases in which intraosseous circulation is up to 100 times greater than normal, which can progress to heart failure. It is a slowly evolving disease with deformities and, due to the progressive increase in density, the bones assume a stony consistency. The anatomopathological substrate shows disorder in the bone apposition and resorption mechanisms, demonstrated histologically by the numerical increase in the cement lines that demarcate the increasingly greater apposition bands in the compromised bone. These lines become so evident that they assume a “mosaic arrangement”, with a progressive, disordered numerical increase in osteoclasts and osteoblasts on the margins of the bone beams, which become irregular, interspersed with fibrosis in the inter-trabecular spaces. “Chalk fractures” result from greater bone density and less elasticity, which is why they have a straight line, similar to broken chalk.
2 – Blood Dyscrasias:
In blood dyscrasias such as leukemia or hemolytic diseases such as anemia (sickle cell, spherocytic and Cooley). They are rare, but circulatory disorders can occur, with extensive bone infarctions, causes of pathological bone fractures.
INFLAMMATIONS :
Inflammations in general are divided into two large groups: non-specific , in which the arrangement of the cells does not allow the etiological agent to be identified, and specific , or granulomatous, in which the cellular arrangement allows the etiology to be identified, as in tuberculosis.
1 – Hematogenous Osteomyelitis:
Hematogenous osteomyelitis stands out among the nonspecific processes, more common in children and adolescents. These mainly affect long bones, most frequently in the metaphyses of the femur and tibia. The location is due to blood stasis in the epiphyseal lines where demand is intense, making it suitable for the development of bacteria. Due to the intensity of the inflammatory process, although infrequent, fractures may occur.
2 – Tuberculosis:
Tuberculosis is a specific process, whose osteolytic lesion can lead to fractures. When located in the spine, Pott’s disease, the disease compromises the intervertebral spaces, with secondary osteolysis that can result in fractures with wedging of the vertebrae with subsequent kyphosis.
3 – Deep Mycoses:
Among the deep mycoses, South American blastomycosis , whose agent is paracoccidioidis brasiliensis , although uncommon in bone location, is the one that most likely causes fractures.
4 – Parasites:
Echinococus Granulosus, among the parasites, is the one that causes most bone fractures . It is a rare disease in our country, known as hydatid cyst. In our experience we had two cases, one of them with a severe osteolytic vertebral lesion and the other, a femoral lesion, which also fractured. Treatment is surgical.
NEOPLASMS:
Regardless of whether they are benign or malignant, tumors can cause fractures, depending on the aggressiveness of the bone structure. Bones with greater overload, such as the vertebrae and those of the lower limb, are more prone to fractures than the others. Benign tumors include osteoblastoma, enchondroma, chondromyxoid fibroma, gigantocellular tumor and hemangioma.
1 – Osteoblastoma:
Osteoblastoma is a neoplasm that is more aggressive, which is why it manifests with osteolysis and can be the cause of fractures, most frequently in long bones or the spine.
2 – Enchondroma:
Enchondroma , which in approximately 50% of cases is present in the phalanges of the hands, although benign and often asymptomatic, can manifest itself as a spontaneous or traumatic fracture. When located in long bones, mainly in the humerus and femur, they can also cause fractures and must be differentiated from bone infarction using imaging methods, sometimes only clarified with a biopsy of the lesion.
3 – Giant Cell Tumor – TGC:
The gigantocellular tumor is most common in the epiphysis of long bones, mainly distal to the femur and proximal to the tibia and humerus. The possibility of fracture arises from the frequency with which it extends to the metaphysis, sometimes with high local aggressiveness.
4 – Chondromyxoid Fibroma:
Chondromyxoid fibroma , most common in the femur and tibia, is slow-growing, eccentric in relation to the bone axis, and can rarely be the cause of fracture.
5 – Hemangioma:
Hemangioma , in long bones or the spine, can be asymptomatic and is sometimes diagnosed by chance finding in a radiographic examination carried out for other reasons. Under certain conditions, however, it manifests itself as a fracture and/or “collapse” of the vertebrae body. It can be isolated or multiple, characterizing bone hemangiomatosis.
6 – Osteosarcoma – Chondrosarcoma:
All primitive malignant bone neoplasms present the possibility of fracture. The ones that most commonly do this are the osteolytic forms of osteosarcoma , mainly teleangectatic.
7 – Malignant hemangioendothelioma:
malignant hemangioendothelioma , due to the intensity of vascularization and resulting intraosseous hemorrhages.
8 – Plasmacytoma / Multiple myeloma:
Osteolytic lesions of plasmacytoma/myeloma , caused by intramedullary clusters of atypical plasma cells, are often the first sign of the disease.
9 – Lymphoma:
Other lytic tumors such as intraosseous lymphomas .
10 – Fibrosarcoma / Malignant Fibrohistiocytoma:
Less common is fibrosarcoma , malignant fibrous histiocytoma.
11 – Liposarcoma:
Liposarcoma , the most common soft tissue tumor, can also present with a fracture .
Bone Metastases – Breast, Prostate, Lung, Kidney and Thyroid Cancer:
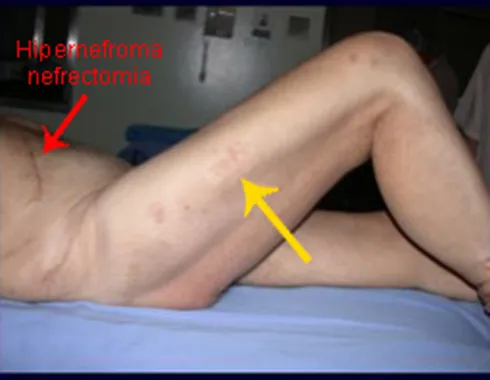
The main manifestation of pathological bone fractures is due to secondary neoplasms or metastases (from the Greek: meta=beyond, stasis=stop). The most frequent bone metastases in men originate from the prostate and lungs. In women, they are of breast and pulmonary origin. Those originating from prostate carcinoma are generally osteocondensant, because, due to the slowness with which the cells reach the vertebrae, through the para-vertebral venous plexus of Batson and in the other bones via arterial blood, the bone tissue reacts with neoformation of inter-trabecular beams that reduce the medullary spaces, in order to condense the bone, clearly evident in imaging methods or pathological examination. As in Paget’s disease, the greater density and less elasticity of bones can cause “chalk line fractures”. Lung metastases, in both sexes, are osteolytic with more frequent involvement of the humerus, pelvis and femur. In the spine, the lesion initially affects the pedicles, while in plasmacytoma/myeloma the involvement predominates in the vertebral body. Breast carcinoma metastases are generally osteolytic. Osteolytic fractures have a pathogenesis based on the greater speed with which cells reach the bone, preventing an adequate osteogenic reaction, contrary to what is observed in prostatic carcinoma. Other neoplasms, originating in the kidney (clear cell carcinoma) and thyroid (follicular carcinoma), due to the intense vascularization that is part of these structures, quickly destroy bone tissue resulting in intensely osteolytic bone fractures, sometimes clinically pulsatile.
Pseudotumor Lesions – Simple Bone Cyst, Aneurysmal Cyst and Non-Ossifying Fibroma:
As for pseudo-neoplastic lesions , the one that most frequently causes fractures is the aneurysmal bone cyst . This process, of unknown etiology, which does not have a cystic appearance and much less is vascular in nature, is known as the benign lesion that has the most aggressive behavior, often simulating malignant neoplasms.
Other pseudo-neoplastic lesions that can fracture are the simple bone cyst of metaphyseal location, when in a bone with greater load such as the femur and tibia, it is prone to fracture. Non-ossifying fibroma , an evolution of the cortical metaphyseal fibrous defect, can also fracture due to its progressive increase in volume, when located in the metaphysis of the femur or tibia.
It is not uncommon for simple or complex bone fractures to hide pathological changes and may result in inadequate orthopedic treatment.
To treat pathological bone fractures, it is necessary to study the entire context that surrounds them. Bone fractures must always be analyzed under multidisciplinary aspects, which take into account the history, age group of the patients, clinical aspects, images, laboratory tests and anatomopathological examination. The joint multidisciplinary study of these data is essential for the diagnosis and management of each case. With the correct diagnosis, the orthopedist will define the treatment. Following what is described in this chapter:
Bone Dysplasias:
1 – Osteopsatirosis or Osteogenesis imperfecta.
Treatment : Clinical: The use of bisphosphonates is currently used.
Orthopedic: intramedullary osteosyntheses to support aligned growth, with telescoped rods, associated or not with osteotomies to correct deformities.
2 – Osteopetrosis
Treatment : Clinical: Prevention of deformities
Orthopedic: osteosynthesis of fractures
3 – Fibrous dysplasia, mono or polyostotic,
Treatment : Clinical: The use of bisphosphonates may have an effect.
Orthopedic: osteosynthesis
Metabolic Changes:
1 – Osteoporosis
Treatment : Clinical: prevention of fractures, avoid caffeine, walking. The use of bisphosphonates may be indicated.
Orthopedic: osteosynthesis of fractures
2 – Osteomalacia and rickets
Treatment : Clinical: Correction of homeostasis, vitamin D, prevention of deformities.
Orthopedic: osteosynthesis of fractures.
3 – Hyperparathyroidism
Treatment : Clinical: resection of the parathyroid tumor and compensation of the metabolic condition, taking into account the marked hypocalcemia that occurs after surgery, as the bone tissue begins to quickly compensate for the existing bone demineralization. Protein supply is essential for the creation of the bone matrix.
Orthopedic: osteosynthesis of fractures, which heal quickly, as the bone is starved for calcium.
Degenerative Diseases:
1 – Eosinophilic granuloma
Treatment : Clinical: corticosteroid therapy
Orthopedic: curettage of the bone lesion. In Calvé’s flat vertebra, the wedging fracture itself leads to healing of the process. In children and adolescents, the vertebral body spontaneously grows, correcting the deformity.
2 – Gaucher disease
Treatment : Clinical:
Orthopedic:
Circulatory disorders:
1 – Paget’s disease
Treatment : Clinical: Bisphosphonates and anti-inflammatories.
Orthopedic: osteosynthesis of fractures
2 – In blood dyscrasias
Treatment : Clinical:
Orthopedic:
Inflammations:
1 – Hematogenous osteomyelitis
Treatment : Clinical: antibiotic therapy
Orthopedic: drainage of abscesses, removal of bone sequestra and stabilization of fractures.
2 – Tuberculosis
Treatment : Clinical: triple regimen for tuberculosis.
Orthopedic: cleaning of caseous abscesses and immobilization, with arthrodesis of the affected joints and osteosynthesis of fractures often being indicated.
3 – South American Blastomycosis , whose agent is paracoccidioidis brasiliensis and
Treatment : Clinical: specific drug treatment for ringworm
Orthopedic: surgical cleaning and specific care for each case.
4 – Echinococcosis, in the form of a hydatid cyst, must be treated surgically.
Neoplasms:
1 – Benign primitives : Orthopedic treatment can include intralesional curettage, local adjuvant, reconstruction with osteosynthesis and autologous graft or methyl methacrylate.
2 – Malignant Primitives : May require neoplasia-oriented chemotherapy treatment and surgical treatment of resection and reconstruction with endoprostheses or biological reconstruction, if possible, or ablative surgery.
3 – Secondary to metastases : Restoring function is essential for the patient’s quality of life. The treatment option for these fractures requires some consideration to make the appropriate choice for each patient. Some of them are subjective, as we have to assume the patient’s likely survival time, clinical possibility that they will fully recover their functions, ability to withstand anesthesia, etc.
We seek to support our decisions on the following parameters:
- Is the injury to the upper limb, lower limb, pelvic girdle or spine?
- Is the injury single or are there multiple injuries?
- Has the fracture already occurred or is there a risk of fracture?
- If no fracture occurred, is 1/3 of the bone’s circumference already compromised? Does the injury cover a long area?
- Did the patient walk before the fracture, did he have normal motor function?
- How long has the patient been treated for the primary disease?
- Are you currently receiving chemotherapy treatment?
- What is this patient’s temporal prognosis?
- What co-morbidities do you have in addition to the neoplasm?
- The type of primary neoplasm responds to radiotherapy
The analysis of these issues will allow a therapeutic decision to be made that assists the patient in recovering their motor function, in line with the treatment of their underlying disease.
Patients with myeloma have a high rate of postoperative infections and usually benefit from local radiotherapy, especially in lesions of the thoracic spine or upper limb, in the first years of the disease. After a few years, when the disease becomes refractory to chemotherapy or bone marrow transplantation, even surgical options are limited due to the intensity of bone involvement.
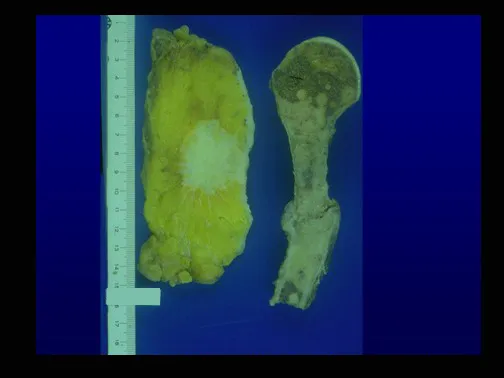
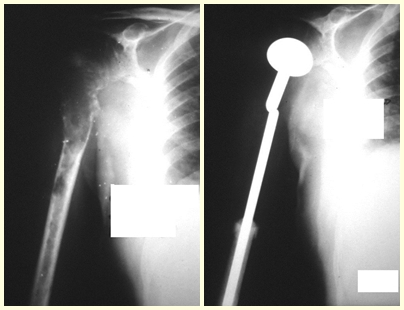
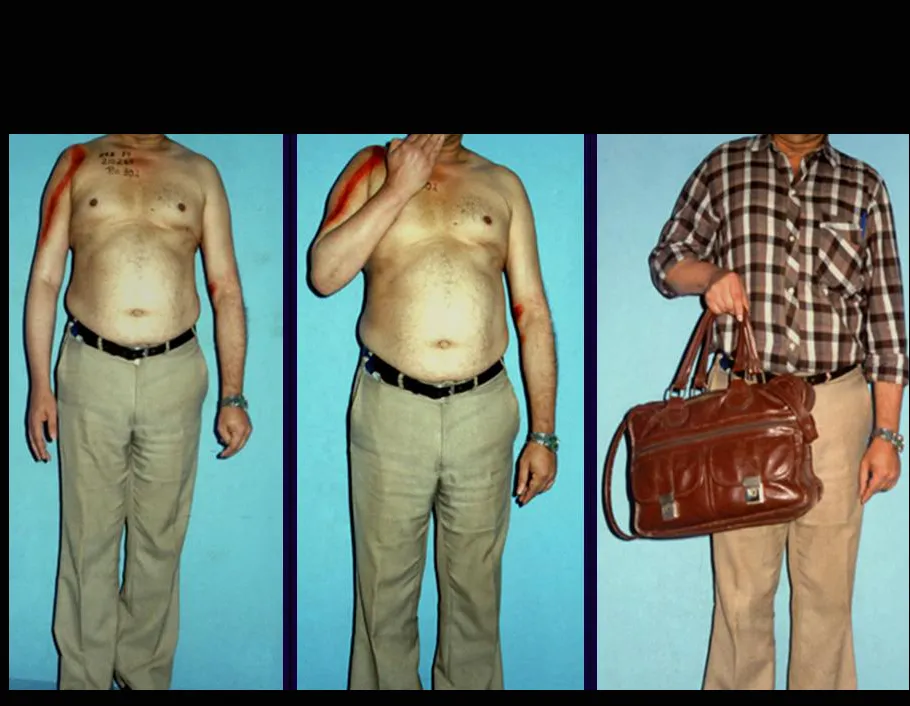
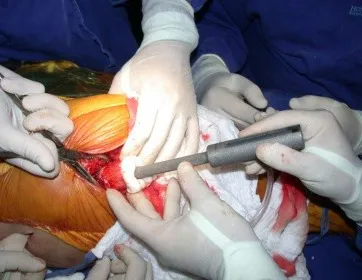
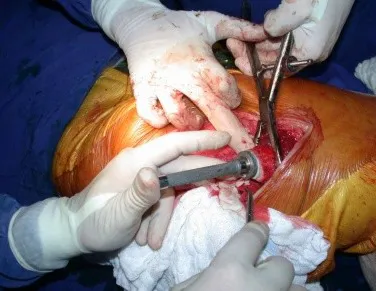
This case in figures 8 to 12 exemplifies a patient with multiple myeloma, presenting an extensive lesion in the proximal half of the right humerus. Despite being myeloma, which responds well to chemotherapy and radiotherapy and even in the upper limb, there is an indication for resection of the lesion and reconstruction with a non-conventional endoprosthesis due to the destruction of the anatomy and providing prompt restoration of function.
Injuries to the lower limb, as it is a load-bearing limb, are best resolved with surgical treatment. The use of palliative radiotherapy, considering a “reserved prognosis”, can cause more suffering when the lesion fractures, as all neoplasms cause the replacement of normal bone tissue by tumor tissue. Therefore, there is bone lysis in all neoplasms, including prostate metastases. It is often mistakenly said that prostate bone metastases are osteoblastic, but what happens pathophysiologically is that in slow-evolving neoplasms there is time for the bone tissue to react to the injury, in an attempt to repair the bone that has been injured, or lysed. if you prefer.
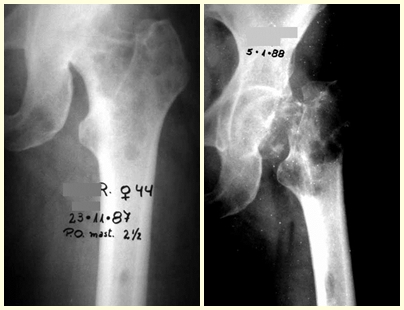
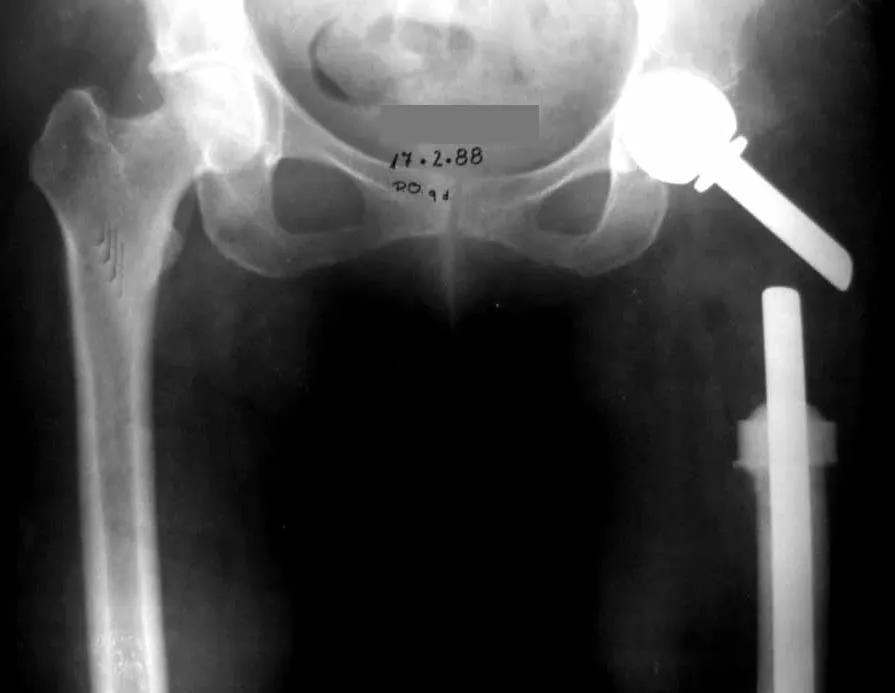
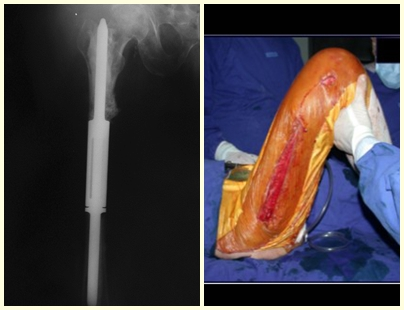
In figures 13 to 19, we illustrate a case of breast cancer metastasis in a patient who had a mastectomy two months ago. We observed numerous lytic lesions in the proximal metaphyseal region of the left femur on December 23, 1987. Local radiotherapy was recommended. The injury did not respond to treatment and in this location, lower limb, in just 40 days the injury progressed and fractured, increasing the patient’s pain and the family’s discomfort.
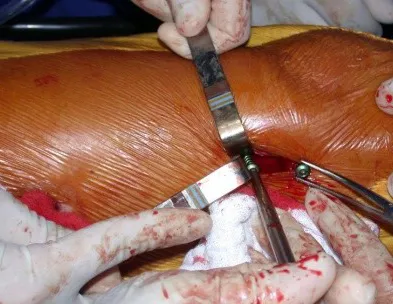
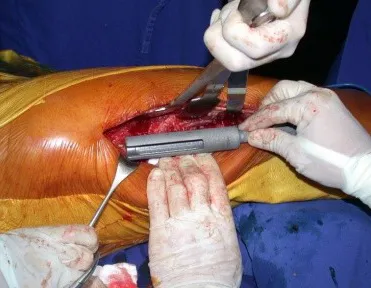
The lesion was resected and replaced with an endoprosthesis.
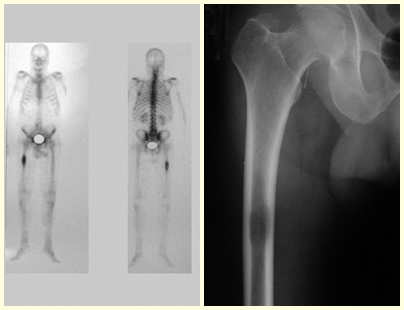
After one year and six months, a lesion appears in the ilio-pubic branch and in the femoral neck on the right side and we should not wait for it to fracture but rather treat it prophylactically.
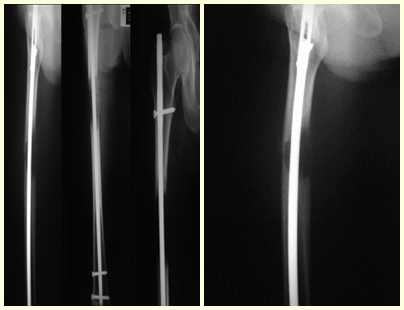
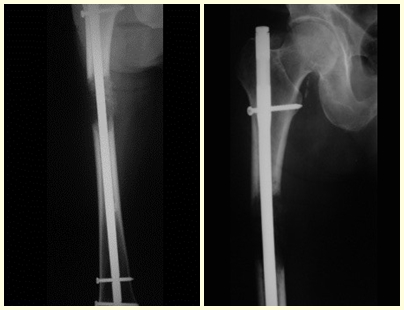
In this last example, a single lesion is observed in the femoral shaft. He was treated with a blocked femoral nail. We observed the progression of the injury each month, complaining of pain and difficulty walking that progressively worsened. Note the local destruction and instability of osteosynthesis, figures 20 to 26.
The patient underwent re-operation, with the locking screws and femoral stem removed, the injured segment resected and reconstructed with a diaphyseal prosthesis.
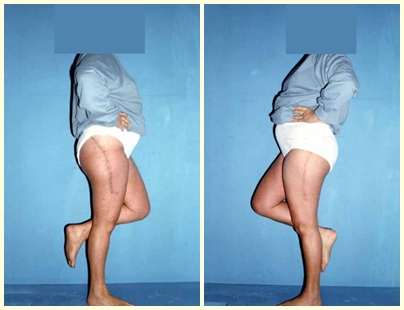
The patient can walk from the first day after surgery, being able to return to their work activity and complementary treatment of the underlying disease.
These examples illustrate the difficulties in approaching pathological fractures and the need for professionals with experience in treating these injuries.
See fracture of the femoral neck in a child due to a simple bone cyst.
See incomplete fracture of the femoral neck in an adult due to renal metastasis.
BIBLIOGRAPHIC REFERENCES:
ALBRIGHT, F., REIFENSTEIN, EC: The parathyroid gland and metabolism of bone disease. Selected studies. Baltimore: William & Wilkins; 1948.
BATSON, OV: The function of the vertebral veins and their role in the spread of metastasis. Ann. Surg., 112:138, 1940.
BRASILEIRO FILHO, G.: Bogliolo pathology. Guanabara Koogan, 7th ed., 2006, pg. 846-847.
DORFMAN, HD & CZERNIAK, B: in Boné tumors, Mosby, St. Louis, USA, 1997, pg.194- 204.
GALASKO, CBS & BENNET, A.: Mechanism of lytic and blastic metastasis diseases of bone. Clin Orthop, 169:20, 1982.
GORHAN, LV. & WEST, WT.: Circulatory changes in osteolytic and osteoblastic reactions. Arch Pathol, 78:673, 1964.
JAFFE, HL.: Metabolic, in Degenerative, and Inflammatory diseases of bone and joints. Lea & Febiger, Philadelphia, 1972, pp. 17l- 180.
LICHTENSTEIN, L. : Histiocytosis Path., 56:84, 1953.
PROSPERO, JD., RIBEIRO BAPTISTA, PP, AMARY, MFA, & CREM DOS SANTOS, P.: Parathyroids: structure, functions and pathology. Acta Orthop. Brasil., 17:2, 2009.
PROSPERO, JD. in Bone Tumors. Ed. Roca, São Paulo, Brazil, 2001, pgs.211-226
RUBIN, P: Dynamic classification of bone dysplasias, Year Book Medical publisher Inc. Osteogenesis imperfecta pg.322 -324
RUBIN, P.: Dynamic classification of bone dysplasia, Year Book Medical publishers Inc., 1964. Osteopetrosis pg. 258 – 280
Author: Prof. Dr. Pedro Péricles Ribeiro Baptista
Orthopedic Oncosurgery at the Dr. Arnaldo Vieira de Carvalho Cancer Institute
Office : Rua General Jardim, 846 – Cj 41 – Cep: 01223-010 Higienópolis São Paulo – SP
Phone: +55 11 3231-4638 Cell:+55 11 99863-5577 Email: drpprb@gmail.com